Introduction
- How do serotonin-blocking drugs work?
- What are the different types of serotonin-blocking drugs?
- What are serotonin-blocking drugs used for?
- What are the side effects of using serotonin-blocking drugs?
- Do serotonin-blocking drugs interact with other drugs?
Introduction
5-hydroxytryptamine receptor antagonists (5-HT3 RAs) are a group of drugs which are used to control nausea and vomiting. They get their name through their ability to block 5-hydroxytryptamine (also known as serotonin) from activating nerves that bring about the vomiting reflex. 5-HT3 RAs, or serotonin blockers, were originally discovered in the 1990s and are one of the newest types of anti-vomiting drugs on the market. The effectiveness of these drugs has revolutionised the management of nausea and vomiting, particularly in individuals undergoing chemotherapy or radiotherapy and in the treatment of nausea and vomiting after surgery.
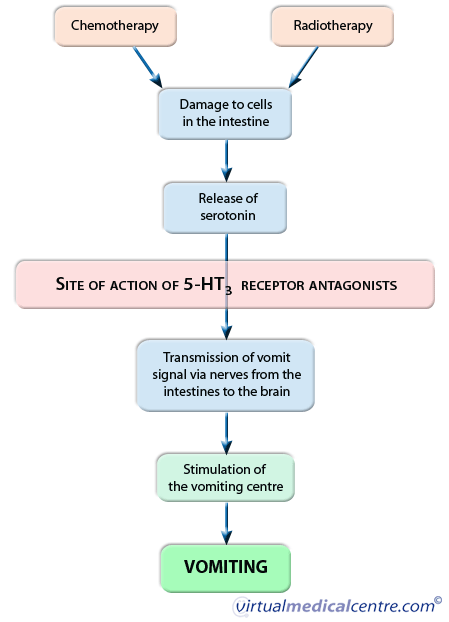
There are currently five serotonin blockers marketed in Australia for treating or preventing chemotherapy-induced nausea and vomiting (CINV), including dolasetron (Anzemet), granisetron (Kytril), ondansetron (Zofran), tropisetron (Navoban) and palonosetron (Aloxi). These drugs work by blocking the action of serotonin on nerves that transmit vomiting impulses from the intestines to the brain.
Despite having different chemical make-ups and different absorption profiles, most drugs in this class are equally effective. The above serotonin blockers are very well tolerated with minimal side effects. These drugs are generally safe to take with other prescription medications, but it is always wise to discuss possible interactions with your doctor or pharmacist before taking two drugs at the same time.
How do serotonin-blocking drugs work?
Vomiting and nausea are brought about by a complex pathway that involves several steps. Ultimately this pathway involves activation of an area within the brain known as the “vomiting centre”. This centre is responsible for co-ordinating signals from the body, processing them, and activating the vomiting reflex. The vomiting centre receives signals from different parts of the brain, including the balance centre, in addition to the stomach and throat.
Serotonin (5-HT3) is the chemical messenger responsible for transmitting vomiting signals. It does this by binding to receptors located on nerves in the stomach and the brain, which transport impulses to the vomiting centre.
Chemotherapy and radiotherapy are thought to bring about vomiting by damaging the cells lining the intestines. These cells then release the serotonin stored inside them, which subsequently binds to nerves that transmit a vomit-inducing impulse from the stomach to the vomiting centre. Once this signal reaches the vomiting centre, serotonin activates other nerves involved in activating the vomit reflex.
Nausea and vomiting after surgery are less well understood, but are likely to result from a number of different factors including the anaesthetic used, the type and duration of surgery, and individual-related factors. Of these, certain types of anaesthetics are thought to be the strongest risk factor.
 |
For more information on the stomach, throat and intestines, see Anatomy of the Gastrointestinal System. |
What are the different types of serotonin-blocking drugs?
First-generation serotonin blockers
Dolasetron, granisetron, ondansetron and tropisetron are called first-generation serotonin blockers. Despite having different chemical structures and absorption by the body, all first-generation drugs work in the same way and have similar side effects.
A huge amount of research has been done on the first-generation serotonin blockers, resulting in the following key points:
- The different serotonin blocking drugs are equally effective and can therefore be used interchangeably;
- These drugs are well tolerated and have few side effects;
- The lowest once-daily dose that fully prevents nausea and vomiting should be used; and
- The tablet form of the drug (oral form) is just as effective in preventing vomiting as the intravenous form.
Second-generation serotonin blockers
Palonosetron was approved by the FDA in 2003 as a second-generation serotonin blocker. It binds much more strongly to its receptor than the first-generation drugs, and therefore works over a longer period of time. Whereas the first-generation drugs are thought to only bind to nerves in the intestines, palonosetron is thought to also work by binding directly to receptors in the brain.
Palonosetron is the first drug to be approved by FDA for prevention of both acute and delayed CINV.
What are serotonin-blocking drugs used for?
Chemotherapy-induced nausea and vomiting (CINV)

CINV can be classified into acute, delayed, anticipatory, breakthrough or refractory, based on when it occurs in relation to the administration of chemotherapy.
CINV affects 70–80% of people with cancer, and has a significant impact on the individual as well as healthcare resources.
Factors which may make you more likely to experience CINV can be broadly categorised into drug-related factors and individual-related factors. Drug-related risk factors include how likely the chemotherapy agent is to induce vomiting (its “emetogenicity”), the dose of the drug and how the drug is taken (e.g. taken by the mouth or directly injected into a vein). Individual-related risk factors include age, gender, previous chemotherapy, history of motion sickness or nausea with pregnancy, and history of heavy alcohol use.
Current guidelines developed as a result of extensive research advocate the use of serotonin blockers in the treatment of both acute and delayed CINV. For the prevention of acute nausea and vomiting with highly vomit-inducing chemotherapy, a combination of a serotonin blocker, a corticosteroid (such as dexamethasone) and substance P (NK-1) blocker (aprepitant) is recommended. These agents should be given before chemotherapy is started.
For the prevention of acute CINV with moderately vomit-inducing chemotherapy, a combination of a serotonin blocker and dexamethasone is recommended, with or without a substance P blocker. It is worth noting that while corticosteroids such as dexamethasone play a useful additional role in preventing vomiting, they have significant side effects. One study found that some individuals receiving dexamethasone for moderately vomit-inducing chemotherapy experienced moderate to severe sleeplessness, stomach discomfort or indigestion, agitation, increased appetite, weight gain or acne in the week following chemotherapy.
Palonosetron is the preferred serotonin blocker for preventing acute nausea and vomiting after moderately vomit-inducing chemotherapy. A single dose of palonosetron is administered before the commencement of chemotherapy, with no need for additional dosing on days 2 and 3.
The most recent guidelines advocate the use of palonosetron injected into the vein in the treatment of delayed CINV on day 1 of chemotherapy only. It is the only serotonin blocker that is available for treating delayed vomiting. The effect of additional doses of palonosetron is not yet completely known.
Your oncologist will evaluate your chemotherapy regimen and your specific risk factors before determining what the best anti-vomiting treatment is for you.
 |
For more information on CINV, see Chemotherapy-Induced Nausea and Vomiting (CINV). |
Radiation-induced nausea and vomiting (RINV)

The prevention of RINV depends on which part of the body is to be irradiated and whether or not the radiation is combined with chemotherapy. When radiotherapy is combined with chemotherapy, the strength of the chemotherapy drugs should determine which anti-vomiting drugs are used. Serotonin-blocking drugs are the preferred anti-vomiting drugs for preventing RINV. Individuals receiving radiation to the upper stomach area and whole upper body should receive a serotonin blocker (ondansetron or granisetron) with or without a corticosteroid (dexamethasone).
Your oncologist will evaluate your radiotherapy regimen and your specific risk factors before determining what the best anti-vomiting treatment is for you.
Post-operative nausea and vomiting (PONV)
Despite developments in anti-vomiting drugs, vomiting and nausea still occurs in 20–30% of individuals after an operation. Risk factors for the development of PONV can be broadly divided into those related to the anaesthetic, those related to the individual, and those related to the type and duration of surgery.
Serotonin-blocking drugs are strongly recommended in the prevention and treatment of PONV. Prevention of nausea and vomiting in these individuals is guided by risk factors that group individuals into high, moderate or low risk of developing PONV. Individuals who are at moderate or high risk of developing PONV based on their risk factors should receive a serotonin blocker in addition to other anti-vomiting drugs. Serotonin-blocking drugs work best when given at the end of surgery.
For people with PONV who did not receive a preventative dose of 5-HT3 RA at the end of surgery, the best treatment is a small dose of a serotonin-blocking drug. Those individuals who develop PONV within the first 6 hours of surgery should be treated with a drug from a different class to that used to prevent the nausea and vomiting. Individuals who develop PONV more than 6 hours after their operation can be treated with any of the anti-vomiting drugs, with the exception of dexamethasone.
Your surgeon or anaesthesiologist will evaluate your specific risk factors before determining what the best anti-vomiting treatment is for you.
What are the side effects of using serotonin-blocking drugs?

Changes in the electrical activity of the heart as recorded on an electrocardiogram (ECG) have been reported with this class of drugs. However, no serious events relating to heart activity have been attributed to this class of drugs. When discussing the use of serotonin-blocking drugs with your physician, you should inform them about any past or existing heart condition you had or have.
There is only limited information on the use of these drugs in pregnant or breastfeeding women, and you should consult your doctor if you intend on becoming pregnant or breastfeeding.
Do serotonin-blocking drugs interact with other drugs?
All of the drugs in this group are processed using the same pathway within the liver. This has the potential to affect the absorption of other drugs which use the same pathway. This risk is greater in individuals receiving chemotherapy and radiotherapy, as well as individuals who are hospitalised. Although these interactions have the potential to affect the absorption of other drugs, no such cases have been reported. It is critical that you tell your physician about all medications you take so that they can evaluate the risk of possible drug interactions.
More information
 |
For more information about nausea and vomiting, including what causes it, how it happens, consequences and managing and treating it, see Nausea and Vomiting (Emesis). |
 |
For more information about drugs, including an introduction to pharmacology, generic versus branded drugs, and the meaning of drug schedules and pregnancy categories, see Drugs. |
References
- Gregory RE, Ettinger DS. 5-HT3 receptor antagonists for the prevention of chemotherapy-induced nausea and vomiting. A comparison of their pharmacology and clinical efficacy. Drugs. 1998;55(2):173-89. [Abstract]
- Hesketh PJ. Chemotherapy-induced nausea and vomiting. N Engl J Med. 2008;358(23):2482-94. [Abstract]
- de Boer-Dennert M, de Wit R, Schmitz PI, et al. Patient perceptions of the side-effects of chemotherapy: The influence of 5HT3 antagonists. Br J Cancer. 1997;76(8):1055-61. [Abstract | Full text]
- 5HT3 receptors [online]. Adelaide, SA: Australian Medicines Handbook; January 2011 [cited 30 April 2011]. Available from: URL link
- Navari RM. Pharmacological management of chemotherapy-induced nausea and vomiting: Focus on recent developments. Drugs. 2009;69(5):515-33. [Abstract]
- Rubenstein EB. Palonosetron: A unique 5-HT3 receptor antagonist indicated for the prevention of acute and delayed chemotherapy-induced nausea and vomiting. Clin Adv Hematol Oncol. 2004;2(5):284-9. [Abstract | Full text]
- Dilly S. Granisetron (Kytril) clinical safety and tolerance. Semin Oncol. 1994;21(3 Suppl 5):10-4. [Abstract]
- Baker PD, Morzorati SL, Ellett ML. The pathophysiology of chemotherapy-induced nausea and vomiting. Gastroenterol Nurs. 2005;28(6):469-80. [Abstract]
- Maricq AV, Peterson AS, Brake AJ, et al. Primary structure and functional expression of the 5HT3 receptor, a serotonin-gated ion channel. Science. 1991;254(5030):432-7. [Abstract]
- Kovac AL. Prevention and treatment of postoperative nausea and vomiting. Drugs. 2000;59(2):213-43. [Abstract]
- Billio A, Morello E, Clarke MJ. Serotonin receptor antagonists for highly emetogenic chemotherapy in adults. Cochrane Database Syst Rev. 2010;(1):CD006272. [Abstract | Full text]
- Jordan K, Sippel C, Schmoll HJ. Guidelines for antiemetic treatment of chemotherapy-induced nausea and vomiting: Past, present, and future recommendations. Oncologist. 2007;12(9):1143-50. [Abstract | Full text]
- Perwitasari DA, Gelderblom H, Atthobari J, et al. Anti-emetic drugs in oncology: pharmacology and individualization by pharmacogenetics. Int J Clin Pharm. 2011;33(1):33-43. [Abstract | Full text]
- Rittenberg CN. The next generation of chemotherapy-induced nausea and vomiting prevention and control: A new 5-HT3 antagonist arrives. Clin J Oncol Nurs. 2004;8(3):307-8, 310. [Abstract]
- Ettinger D, Armstrong K, Barbour S, et al. NCCN clinical practice guidelines in oncology: Antiemesis [online]. Fort Washington, PA: National Comprehensive Cancer Care Network; 2011 [cited 30 May 2011]. Available from: URL link
- Liau CT, Chu NM, Liu HE, et al. Incidence of chemotherapy-induced nausea and vomiting in Taiwan: Physicians’ and nurses’ estimation vs. patients’ reported outcomes. Support Care Cancer. 2005;13(5):277-86. [Abstract]
- Lohr L. Chemotherapy-induced nausea and vomiting. Cancer J. 2008;14(2):85-93. [Abstract]
- Pollera CF, Giannarelli D. Prognostic factors influencing cisplatin-induced emesis. Definition and validation of a predictive logistic model. Cancer. 1989;64(5):1117-22. [Abstract]
- Roila F, Herrstedt J, Aapro M, et al. Guideline update for MASCC and ESMO in the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting: Results of the Perugia consensus conference. Ann Oncol. 2010;21(Suppl 5):v232-43. [Abstract | Full text]
- Prevention of acute/delayed nausea and vomiting (emesis) [online]. Bethesda, MD: National Cancer Institute; 16 March 2011 [cited 30 May 2011]. Available from: URL link
- Grunberg SM, Warr D, Gralla RJ, et al. Evaluation of new antiemetic agents and definition of antineoplastic agent emetogenicity – state of the art. Support Care Cancer. 2011;19(Suppl 1):S43-7. [Abstract]
- Vardy J, Chiew KS, Galica J, et al. Side effects associated with the use of dexamethasone for prophylaxis of delayed emesis after moderately emetogenic chemotherapy. Br J Cancer. 2006;94(7):1011-5. [Abstract | Full text]
- Salvo N, Doble B, Khan L, et al. Prophylaxis of radiation-induced nausea and vomiting using 5-hydroxytryptamine-3 serotonin receptor antagonists: A systematic review of randomised trials. Int J Radiat Oncol Biol Phys. 2010 Nov 13. [Abstract]
- Cohen MM, Duncan PG, DeBoer DP, Tweed WA. The postoperative interview: Assessing risk factors for nausea and vomiting. Anesth Analg. 1994;78(1):7-16. [Abstract | Full text]
- Gan TJ, Meyer T, Apfel CC, et al. Consensus guidelines for managing postoperative nausea and vomiting. Anesth Analg. 2003;97(1):62-71. [Abstract | Full text]
- Keefe DL. The cardiotoxic potential of the 5-HT(3) receptor antagonist antiemetics: Is there cause for concern? Oncologist. 2002(7):65-72. [Abstract | Full text]
- Aapro M, Blower P. 5-hydroxytryptamine type-3 receptor antagonists for chemotherapy-induced and radiotherapy-induced nausea and emesis: Can we safely reduce the dose of administered agents? Cancer. 2005;104(1):1-18. [Abstract | Full text]
- Navari RM, Koeller JM. Electrocardiographic and cardiovascular effects of the 5-hydroxytryptamine3 receptor antagonists. Ann Pharmacother. 2003;37(9):1276-86. [Abstract]
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.