PSA Testing (Prostate Specific Antigen Testing)
- What is prostate specific antigen (PSA)?
- When is PSA testing used?
- Benefits and risks of PSA testing
- Limitations of PSA testing
- How to prepare for PSA testing
- How is PSA tested?
- What do the results mean?
- My PSA test was abnormal. What next?
What is prostate cancer?
The prostate is a gland found only in men. It produces fluid that forms part of the semen which is expelled when a man ejaculates. The gland wraps around the urethra (the tube in the penis through which urine and semen passes through) at the base of the bladder. In healthy men, the prostate is slightly larger than a walnut. Cancer may develop within the cells of the prostate gland; this is termed ‘prostate cancer‘.
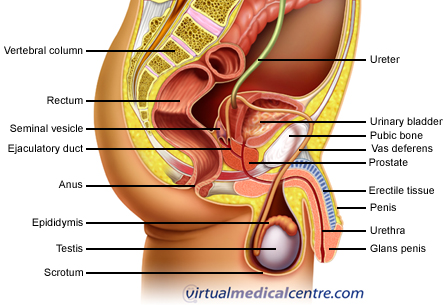
Prostate cancer is the most common form of cancer in Australian men (after non-melanoma skin cancer). One in six men will develop prostate cancer at some point in their lives. One in 34 will die from it, making it the second leading cause of death in Australian men.
What is prostate specific antigen?
Prostate specific antigen (PSA) is a protein that is produced by the cells of the prostate gland and enters the bloodstream. A range of disorders involving the prostate cause its cells to produce more PSA. This raised amount of PSA can then be measured with a blood test. Raised PSA levels can indicate several different disorders involving the prostate, one of which is prostate cancer.
When is PSA testing used?
 PSA testing is used to identify the early stages of prostate cancer. It is currently one of the few widely used tests for prostate cancer available. The other examination used to detect this cancer is a digital rectal examination (DRE), in which a doctor inserts a gloved finger into the rectum (back passage) to feel the prostate and determine if it is enlarged or if any lumps are present. PSA and DRE are generally performed together to check for prostate cancer.
PSA testing is used to identify the early stages of prostate cancer. It is currently one of the few widely used tests for prostate cancer available. The other examination used to detect this cancer is a digital rectal examination (DRE), in which a doctor inserts a gloved finger into the rectum (back passage) to feel the prostate and determine if it is enlarged or if any lumps are present. PSA and DRE are generally performed together to check for prostate cancer.
The Cancer Council Australia does not currently recommend that all men undergo routine screening for prostate cancer. Studies have shown that this approach does not reduce the number of men who die from prostate cancer. Although early identification of prostate cancer is very important, there are several significant limitations and disadvantages of PSA testing that restrict its use. PSA testing is therefore done on a ‘case by case’ basis. Men who are at higher risk of developing prostate cancer, or men who have symptoms that may indicate cancer, are more likely to benefit from testing.
The main risk factor for prostate cancer is age. PSA testing is not generally carried out on men aged under 50. Around 5–10% of prostate cancer is due to genetic defects, so men who have a family history of prostate cancer are more at risk of developing it themselves. Men who have a first degree relative (father, brother or son) who have had prostate cancer have a 2–3 times greater chance of developing prostate cancer themselves than men who don’t. This risk may be higher if the relative was diagnosed before the age of 60. Other men who are at increased risk of prostate cancer are those who have had elevated PSA tests or abnormal prostate biopsies in the past.
Benefits and risks of PSA testing
Early detection of aggressive prostate cancer through PSA testing may allow the disease to be treated early enough that it can be cured. PSA levels may be elevated 5–10 years before prostate cancer produces any symptoms. Early detection may also reduce the risk of complications due to the treatment of the disease, or of the disease itself. This in turn will lead to improved quality of life.
The risks of the testing procedure itself are extremely low. There is a possibility of some pain or bleeding from the site of the blood test.
Risks associated with PSA testing generally relate to the consequences of the results. False positive tests (PSA elevated, but no cancer) may lead to unnecessary investigations or treatment, which can have serious side effects. Prostate biopsy very rarely leads to complications requiring hospitalisation (< 1%), but anxiety and short term pain related to testing are common. Even when a prostate biopsy is negative, significant distress and chronic anxiety may result.
Although the treatment of prostate cancer (removal of the prostate gland, or radiotherapy) very rarely causes death, significant complications include sexual dysfunction, bowel problems and urinary incontinence. Sexual dysfunction may occur in 20–70% of patients, and urinary problems in 15–50% of patients following radical prostatectomy.Radiotherapy may lead to sexual dysfunction in 20–45% of cases, urinary incontinence in 2–16%, and bowel dysfunction in 6–25% of men who were previously normal in these areas.
Some forms of prostate cancer spread so slowly that the disease does not cause health problems, and the man who has it dies of something unrelated. Most men with prostate cancer die with it rather than from it. Treatment of this type of cancer does not change this outcome and could also lead to side effects. In addition, false positive tests or discovery of ‘nonthreatening’ cancer is likely to lead to needless worry and stress.
Limitations of PSA testing
 PSA testing is currently the most widely used method to screen for prostate cancer. Unfortunately, it has several significant limitations. Screening of all men for prostate cancer has not been shown to reduce the number who die from this disease.
PSA testing is currently the most widely used method to screen for prostate cancer. Unfortunately, it has several significant limitations. Screening of all men for prostate cancer has not been shown to reduce the number who die from this disease.
PSA testing is not 100% accurate. Some men who have prostate cancer will not have elevated PSA levels (it is not 100% sensitive). On the other hand, most men who have elevated PSA levels do not turn out to have prostate cancer. Other (noncancerous) conditions that cause the cells of the prostate to produce higher levels of PSA include benign prostatic hyperplasia (BPH), urinary tract infections, and prostatitis. BPH is the most common cause of elevated PSA levels. It is very common for the prostate gland to become larger (hypertrophy) as a man ages. About 80% of men will experience BPH. Symptoms of BPH are caused by the enlarged prostate pressing on the urethra. This results in an altered flow of urine (dribbling, difficulty starting, stop/start urination). BPH is not cancer, and does not lead to cancer. If a man is found to have raised PSA levels, he is likely to undergo further tests to make sure the result is not due to prostate cancer. These tests have significant side effects, and are likely to be ‘unnecessary’ because cancer is not present.
Even if the raised PSA levels are due to cancer, not all forms of prostate cancer develop and progress in the same pattern. The lifetime risk of developing prostate cancer is 1 in 6. The lifetime risk of dying from prostate cancer is 1 in 34. Most men with prostate cancer die of something else. Some men have raised PSA levels because of tiny deposits of cancer in their prostate that will never grow large enough to spread and cause health problems. This is the case in 25–50% of prostate cancer. Identification of this pattern of cancer is likely to result in needless worry for men who choose not to investigate or treat it further. Alternatively, men who choose to undergo further investigation and treatment may suffer significant side effects due to the intervention.
Unfortunately, PSA testing does not identify whether cancer is definitely present, or if it is the type that urgently needs to be treated or the type that is harmless.
How to prepare for PSA testing
- If possible, avoid ejaculating for 48 hours before the test.
- Inform your doctor of medications you are taking before the test, as some may interfere with results.
- If you have recently had a needle biopsy, prostate surgery or a cystoscopy, PSA results may be difficult to interpret and testing should be delayed for several weeks.
- Discuss symptoms such as painful urination, urgency to urinate, or discharge from the penis with your doctor. These may indicate prostatitis or a urinary tract infection.
How is PSA tested?
PSA is detected using a simple blood test. A trained health professional will swab your arm with an antiseptic wipe, then insert a sterile needle into a vein (usually at the front of the elbow) to draw a small sample of blood (about 5 mL). You may feel a ‘scratch-like’ pain during testing. The puncture site will then be covered with a small dressing. This procedure takes 3–5 minutes. The blood is sent in a test tube to a laboratory for testing. It may take about a week to get results of the test back, though this varies according to the facility. Your doctor will discuss the results with you.
What do the results mean?
PSA levels are considered ‘normal’ for men older than 40 if it is below 4 ng/mL (nanograms per millilitre). Men younger than 40 should ideally have a PSA below 2.7ng/mL. PSA levels are considered ‘elevated’ if it is above 4 ng/mL.
- 25% of men with PSA levels of 4–10 ng/mL have prostate cancer
- 42–64% of men with PSA levels > 10gn/mL have prostate cancer
If the PSA test is normal, there is no urgency for a follow up test. Discuss with your doctor how long to wait until another PSA test is required.
PSA may occur in the bloodstream in a ‘free’ form, or it may be bound to proteins. Normally, PSA is produced by the prostate cells; it then goes through a series of changes where it is activated, then inactivated. The inactivated form enters the bloodstream and circulates on its own (as free PSA). A small amount of the active PSA enters the circulation without this full process occurring. The active PSA is quickly bound to proteins in the blood, and circulates in a ‘bound’ or ‘complexed’ form.
Prostate cancer is associated with a lower concentration of free PSA in the bloodstream than occurs due to benign conditions such as BPH. Cancer disrupts the normal structure of the prosate cells, resulting in increased amounts of active PSA entering the bloodstream before being inactivated. Thus, the percentage of free (inactivated) PSA in the circulation is reduced, and there is a higher amount of bound PSA.
The ratio of free to total PSA has been used to improve the accuracy of PSA testing for screening for prostate cancer, especially for indeterminate levels (4.1–10 ng/mL). At these levels, if the free to total PSA ratio is low, there is a greater chance that the elevation is due to cancer rather than to a benign process. For example, in this range, the probably of cancer is 56% when 10% of total PSA is free, versus 8% if more than 25% of PSA is free.
The best free to total PSA ratio for distinguishing between prostate cancer and other causes of elevated PSA is unknown. It has been suggested that the use of free PSA levels in determining the need for a biopsy is only clinically useful at ‘extreme’ levels, though these have not been clearly outlined.Significantly lower levels of free PSA (< 14%) may also be associated with more aggressive forms of prostate cancer, though the application of this finding requires further study.
Free PSA needs to be specifically requested on a laboratory form (“PSA free”), and will only be performed by some laboratories if the total PSA level is between 4 and 10 ng/mL. The total PSA is requested and reported separately, and the percentage of free PSA is provided. Many laboratories report that, for total PSA levels of 4–10 ng/mL and if free/total PSA is less than 10%, prostate cancer is likely. At >25% free/total PSA, BPH is more likely.
My PSA test was abnormal. What next?
Most men who decide to have their PSA levels tested will generally already have decided to undergo further investigations if the result is abnormal. Keep in mind that the majority of men with elevated PSA do not have cancer.
If a PSA level is high for a man’s age, the PSA test will generally be repeated. If it is still found to be elevated, a transrectal ultrasound may be performed. Generally the next stage in testing is a prostate biopsy. Further treatment will depend on the results of the biopsy.
If prostate cancer is found, treatment options include waiting and monitoring the cancer, surgery, radiotherapy, hormonal medication, and other treatments. Factors such as the location of the cancer, how aggressive it is (the Gleason score), the man’s age, and other medical factors will be taken into account in determining the most appropriate treatment.
More information
 | For more information on prostate cancer, including diagnoses, types of treatments, and some useful tools, videos and animations, see Prostate Cancer. |
References
- Ries LAG, Melbert D, Krapcho M, et al (eds). SEER Cancer Statistics Review, 1975-2004 Based on November 2006 SEER data submission [online]. Bethesda, MD: National Cancer Institute; 2007 [cited 17 September 2008]. Available from: URL link
- Dorr VJ, Williamson SK, Stephens RL. An evaluation of prostate-specific antigen as a screening test for prostate cancer. Arch Intern Med. 1993;153(22):2529-37. [Abstract | Full text]
- Gann PH, Hennekens CH, Stampfer MJ. A prospective evaluation of plasma prostate-specific antigen for detection of prostatic cancer. JAMA. 1995;273(4):289-94. [Abstract | Full text]
- Stamey TA, Yang N, Hay AR, et al. Prostate-specific antigen as a serum marker for adenocarcinoma of the prostate. N Engl J Med. 1987;317(15):909-16. [Abstract]
- Tchetgen MB, Song JT, Strawderman M, et al. Ejaculation increases the serum prostate-specific antigen concentration. Urology. 1996;47(4):511-6. [Abstract]
- Mettlin C, Jones G, Averette H, et al. Defining and updating the American Cancer Society guidelines for the cancer-related checkup: Prostate and endometrial cancers. CA Cancer J Clin. 1993;43(1):42-6. [Full text]
- American Urological Association. Early detection of prostate cancer and use of transrectal ultrasound. In: American Urological Association 1992 Policy Statement Book. Baltimore: Lippincott Williams & Wilkins; 1992. [Book]
- Ries LAG, Eisner MP, Kosary CL, et al. SEER Cancer Statistics Review, 1973-1999 [online]. Bethesda, MD: National Cancer Institute; 2002 [cited 17 September 2008]. Available from: URL link
- Potosky AL, Miller BA, Albertsen PC, Kramer BS. The role of increasing detection in the rising incidence of prostate cancer. JAMA. 1995;273(7):548-52. [Abstract | Full text]
- Whitmore WF Jr. Natural history of low-stage prostatic cancer and the impact of early detection. Urol Clin North Am. 1990;17(4):689-97. [Abstract]
- Lu-Yao GL, Friedman M, Yao SL. Use of radical prostatectomy among Medicare beneficiaries before and after the introduction of prostate specific antigen testing. J Urol. 1997;157(6):2219-22. [Abstract]
- Ciatto S, Bonardi R, Lombardi C, et al. Predicting prostate biopsy outcome by findings at digital rectal examination, transrectal ultrasonography, PSA, PSA density and free-to-total PSA ratio in a population-based screening setting. Int J Biol Markers. 2001;16(3):179-82. [Abstract]
- Harris R, Lohr KN. Screening for prostate cancer: an update of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2002;137(11):917-29. [Abstract | Full text]
- Screening for Prostate cancer: Systematic Evidence Review, No 16 [online]. Rockville, MD: Agency for Healthcare Research and Quality, US Department of Health and Human Services; October 2002 [cited 15 September 2008]. Available from: URL link
- Draisma G, Boer R, Otto SJ, et al. Lead times and overdetection due to prostate-specific antigen screening: Estimates from the European Randomized Study of Screening for Prostate Cancer. J Natl Cancer Inst. 2003;95(12):868-78. [Abstract | Full text]
- Carroll P, Coley C, McLeod D, et al. Prostate-specific antigen best practice policy – Part I: Early detection and diagnosis of prostate cancer. Urology. 2001;57(2):217-24. [Abstract]
- Smith RA, von Eschenbach AC, Wender R, et al. American Cancer Society guidelines for the early detection of cancer: Update of early detection guidelines for prostate, colorectal, and endometrial cancers. Also: Update 2001 – Testing for early lung cancer detection. CA Cancer J Clin. 2001;51(1):38-75. [Abstract | Full text]
- Prostate cancer screening position statement [online]. Surry Hills, NSW: Cancer Council Australia; April 2005 [cited 12 September 2008]. Available from: URL link
- Mettlin C, Lee F, Drago J, Murphy GP. The American Cancer Society National Prostate Cancer Detection Project. Findings on the detection of early prostate cancer in 2425 men. Cancer. 1991;67(12):2949-58. [Abstract | Full text]
- Johns LE, Houlston RS. A systematic review and meta-analysis of familial prostate cancer risk. BJU Int. 2003;91(9):789-94. [Abstract | Full text]
- Liede A, Karlan BY, Narod SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: A review of the literature. J Clin Oncol. 2004;22(4):735-42. [Abstract]
- Roder D. Epidemiology of cancer in Indigenous Australians: Implications for service delivery. Paper presented at the 9th National Rural Health Conference: Standing up for rural health: Learning from the past, action in the future. 7-10 March 2007; Albury, NSW. [Abstract | Full text]
- Punglia RS, D’Amico AV, Catalona WJ, et al. Effect of verification bias on screening for prostate cancer by measurement of prostate-specific antigen. N Engl J Med. 2003;349(4):335-42. [Abstract | Full text]
- Stroumbakis N, Cookson MS, Reuter VE, Fair WR. Clinical significance of repeat sextant biopsies in prostate cancer patients. Urology. 1997;49(3A Suppl):113-8. [Abstract]
- Ellis WJ, Brawer MK. Repeat prostate needle biopsy: Who needs it? J Urol. 1995;153(5):1496-8. [Abstract]
- Levine MA, Ittman M, Melamed J, Lepor H. Two consecutive sets of transrectal ultrasound guided sextant biopsies of the prostate for the detection of prostate cancer. J Urol. 1998;159(2):471-5. [Abstract]
- Eichler K, Hempel S, Wilby J, et al. Diagnostic value of systematic biopsy methods in the investigation of prostate cancer: A systematic review. J Urol. 2006;175(5):1605-12. [Abstract]
- McNaughton Collins M, Ransohoff DF, Barry MJ. Early detection of prostate cancer. Serendipity strikes again. JAMA. 1997;278(18):1516-9. [Abstract]
- Brawer MK, Chetner MP, Beatie J, et al. Screening for prostatic carcinoma with prostate specific antigen. J Urol. 1992;147(3 Pt 2):841-5. [Abstract]
- Catalona WJ, Smith DS, Ratliff TL, Basler JW. Detection of organ-confined prostate cancer is increased through prostate-specific antigen-based screening. JAMA. 1993;270(8):948-54. [Abstract | Full text]
- Crawford ED, DeAntoni EP, Etzioni R, et al. Serum prostate-specific antigen and digital rectal examination for early detection of prostate cancer in a national community-based program. The Prostate Cancer Education Council. Urology. 1996;47(6):863-9. [Abstract]
- Brawer MK. Prostate-specific antigen: Current status. CA Cancer J Clin. 1999;49(5):264-81. [Abstract | Full text]
- Meigs JB, Barry MJ, Oesterling JE, Jacobsen SJ. Interpreting results of prostate-specific antigen testing for early detection of prostate cancer. J Gen Intern Med. 1996;11(9):505-12. [Abstract | Full text]
- Catalona WJ, Richie JP, Ahmann FR, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: Results of a multicenter clinical trial of 6,630 men. J Urol. 1994;151(5):1283-90. [Abstract]
- Schröder FH, van der Cruijsen-Koeter I, de Koning HJ, et al. Prostate cancer detection at low prostate specific antigen. J Urol. 2000;163(3):806-12. [Abstract]
- Coley CM, Barry MJ, Fleming C, Mulley AG. Early detection of prostate cancer. Part I: Prior probability and effectiveness of tests. The American College of Physicians. Ann Intern Med. 1997;126(5):394-406. [Abstract]
- Polascik TJ, Oesterling JE, Partin AW. Prostate specific antigen: A decade of discovery – what we have learned and where we are going. J Urol. 1999;162(2):293-306. [Abstract]
- Bretton PR. Prostate-specific antigen and digital rectal examination in screening for prostate cancer: A community-based study. South Med J. 1994;87(7):720-3. [Abstract | Full text]
- Muschenheim F, Omarbasha B, Kardjian PM, Mondou EN. Screening for carcinoma of the prostate with prostate specific antigen. Ann Clin Lab Sci. 1991;21(6):371-80. [Abstract]
- Yamamoto T, Ito K, Ohi M, Kubota Y. Diagnostic significance of digital rectal examination and transrectal ultrasonography in men with prostate-specific antigen levels of 4 NG/ML or less. Urology. 2001;58(6):994-8. [Abstract]
- Whittemore AS, Cirillo PM, Feldman D, Cohn BA. Prostate specific antigen levels in young adulthood predict prostate cancer risk: Results from a cohort of Black and White Americans. J Urol. 2005;174(3):872-6. [Abstract]
- Essink-Bot ML, de Koning HJ, Nijs HG, et al. Short-term effects of population-based screening for prostate cancer on health-related quality of life. J Natl Cancer Inst. 1998;90(12):925-31. [Abstract | Full text]
- McNaughton-Collins M, Fowler FJ Jr, Caubet JF, et al. Psychological effects of a suspicious prostate cancer screening test followed by a benign biopsy result. Am J Med. 2004;117(10):719-25. [Abstract | Full text]
- Begg CB, Riedel ER, Bach PB, et al. Variations in morbidity after radical prostatectomy. N Engl J Med. 2002;346(15):1138-44. [Abstract | Full text]
- Lu-Yao GL, Albertsen P, Warren J, Yao SL. Effect of age and surgical approach on complications and short-term mortality after radical prostatectomy – A population-based study. Urology. 1999;54(2):301-7. [Abstract]
- Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: The Prostate Cancer Outcomes Study. JAMA. 2000;283(3):354-60. [Abstract | Full text]
- Rietbergen JB, Kruger AE, Kranse R, Schroder FH. Complications of transrectal ultrasound-guided systematic sextant biopsies of the prostate: Evaluation of complication rates and risk factors within a population-based screening program. Urology. 1997;49(6):875-80. [Abstract]
- Fowler FJ Jr, Barry MJ, Walker-Corkery B, et al. The impact of a suspicious prostate biopsy on patients’ psychological, socio-behavioral, and medical care outcomes. J Gen Intern Med. 2006;21(7):715-21. [Abstract | Full text]
- Johansson JE, Holmberg L, Johansson S, et al. Fifteen-year survival in prostate cancer. A prospective, population-based study in Sweden. JAMA. 1997;277(6):467-71. [Abstract | Full text]
- Johansson JE, Andrén O, Andersson SO, et al. Natural history of early, localized prostate cancer. JAMA. 2004;291(22):2713-9. [Abstract | Full text]
- Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA. 2005;293(17):2095-101. [Abstract | Full text]
- Hamilton AS, Stanford JL, Gilliland FD, et al. Health outcomes after external-beam radiation therapy for clinically localized prostate cancer: Results from the Prostate Cancer Outcomes Study. J Clin Oncol. 2001;19(9):2517-26. [Abstract | Full text]
- Ilic D, O’Connor D, Green S, Wilt T. Screening for prostate cancer. Cochrane Database Syst Rev. 2006;3:CD004720. [Abstract | Full text]
- Gohagan JK, Prorok PC, Hayes RB, Kramer BS. The Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial of the National Cancer Institute: History, organization, and status. Control Clin Trials. 2000;21(6 Suppl):251S-272S. [Abstract]
- Schroder FH. The European Screening Study for Prostate Cancer. Can J Oncol. 1994;4(Suppl 1):102-5.
- Tchetgen MB, Oesterling JE. The effect of prostatitis, urinary retention, ejaculation, and ambulation on the serum prostate-specific antigen concentration. Urol Clin North Am. 1997;24(2):283-91. [Abstract]
- Nadler RB, Humphrey PA, Smith DS, et al. Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J Urol. 1995;154(2 Pt 1):407-13. [Abstract]
- Etzioni R, Penson DF, Legler JM, et al. Overdiagnosis due to prostate-specific antigen screening: Lessons from U.S. prostate cancer incidence trends. J Natl Cancer Inst. 2002;94(13):981-90. [Abstract | Full text]
- Yuan JJ, Coplen DE, Petros JA, et al. Effects of rectal examination, prostatic massage, ultrasonography and needle biopsy on serum prostate specific antigen levels. J Urol. 1992;147(3 Pt 2):810-4. [Abstract]
- Chybowski FM, Bergstralh EJ, Oesterling JE. The effect of digital rectal examination on the serum prostate specific antigen concentration: Results of a randomized study. J Urol. 1992;148(1):83-6. [Abstract]
- Andruss CM, Dinella TJ, Macpherson DS, et al. Effect of digital rectal examination on serum prostate-specific antigen in a primary care setting. Arch Intern Med. 1995;155(4):389-92. [Abstract]
- Herschman JD, Smith DS, Catalona WJ. Effect of ejaculation on serum total and free prostate-specific antigen concentrations. Urology. 1997;50(2):239-43. [Abstract]
- Kawakami J, Siemens DR, Nickel JC. Prostatitis and prostate cancer: Implications for prostate cancer screening. Urology. 2004;64(6):1075-80. [Abstract]
- Simardi LH, Tobias-Machado M, Kappaz GT, et al. Influence of asymptomatic histologic prostatitis on serum prostate-specific antigen: A prospective study. Urology. 2004;64(6):1098-101. [Abstract]
- Guess HA, Heyse, JF, Gormley GJ. The effect of finasteride on prostate-specific antigen in men with benign prostatic hyperplasia. Prostate. 1993;22(1):31-7. [Abstract]
- Roehrborn CG, Marks LS, Fenter T, et al. Efficacy and safety of dutasteride in the four-year treatment of men with benign prostatic hyperplasia. Urology. 2004;63(4):709-15. [Abstract]
- Andriole GL, Guess HA, Epstein JI, et al. Treatment with finasteride preserves usefulness of prostate-specific antigen in the detection of prostate cancer: Results of a randomized, double-blind, placebo-controlled clinical trial. PLESS Study Group. Proscar Long-term Efficacy and Safety Study. Urology. 1998;52(2):195-201. [Abstract]
- Etzioni RD, Howlader N, Shaw PA, et al. Long-term effects of finasteride on prostate specific antigen levels: Results from the prostate cancer prevention trial. J Urol. 2005;174(3):877-81. [Abstract]
- Labrie F, Candas B, Dupont A, et al. Screening decreases prostate cancer death: First analysis of the 1988 Quebec prospective randomized controlled trial. Prostate. 1999;38(2):83-91. [Abstract | Full text]
- Smith DS, Catalona WJ, Herschman JD. Longitudinal screening for prostate cancer with prostate-specific antigen. JAMA. 1996;276(16):1309-15. [Abstract | Full text]
- Crawford ED, Pinsky PF, Chia D, et al. Prostate specific antigen changes as related to the initial prostate specific antigen: Data from the prostate, lung, colorectal and ovarian cancer screening trial. J Urol. 2006;175(4):1286-90. [Abstract]
- Stamey TA, Johnstone IM, McNeal JE, et al. Preoperative serum prostate specific antigen levels between 2 and 22 ng./ml. correlate poorly with post-radical prostatectomy cancer morphology: Prostate specific antigen cure rates appear constant between 2 and 9 ng./ml. J Urol. 2002;167(1):103-11. [Abstract]
- Barry MJ. Clinical practice. Prostate-specific-antigen testing for early diagnosis of prostate cancer. N Engl J Med. 2001;344(18):1373-7. [Abstract]
- Dunn AS, Shridharani KV, Lou W, et al. Physician-patient discussions of controversial cancer screening tests. Am J Prev Med. 2001;20(2):130-4. [Abstract | Full text]
- Baade PD, Steginga SK, Pinnock CB, Aitken JF. Communicating prostate cancer risk: What should we be telling our patients? MJA. 2005;182(9):472-5. [Abstract | Full text]
- Screening for prostate cancer. American College of Physicians. Ann Intern Med. 1997;126(6):480-4. [Abstract]
- Lilja H, Christensson A, Dahlén U, et al. Prostate-specific antigen in human serum occurs predominantly in complex with alpha-1-antichymotrypsin. Clin Chem. 1991;37(9):1618-25. [Abstract | Full text]
- Mikolajczyk SD, Marks LS, Partin AW, Rittenhouse HG. Free prostate-specific antigen in serum is becoming more complex. Urology. 2002;59(6):797-802. [Abstract]
- Björk T, Piironen T, Pettersson K, et al. Comparison of analysis of the different prostate-specific antigen forms in serum for detection of clinically localized prostate cancer. Urology. 1996;48(6):882-8. [Abstract]
- Christensson A, Björk T, Nilsson O, et al. Serum prostate specific antigen complexed to alpha-1-antichymotrypsin as an indicator of prostate cancer. J Urol. 1993;150(1):100-5. [Abstract]
- Partin AW, Hanks GE, Klein EA, et al. Prostate-specific antigen as a marker of disease activity in prostate cancer. Oncology. 2002;16(8):1024-38. [Abstract | Full text]
- Balk SP, Ko YJ, Bubley GJ. Biology of prostate-specific antigen. J Clin Oncol. 2003;21(2):383-91. [Abstract | Full text]
- Catalona WJ, Partin AW, Slawin KM, et al. Use of the percentage of free prostate-specific antigen to enhance differentiation of prostate cancer from benign prostatic disease: A prospective multicenter clinical trial. JAMA. 1998;279(19):1542-7. [Abstract | Full text]
- Hoffman RM, Clanon DL, Littenberg B, et al. Using the free-to-total prostate-specific antigen ratio to detect prostate cancer in men with nonspecific elevations of prostate-specific antigen levels. J Gen Intern Med. 2000;15(10):739-48. [Abstract | Full text]
- Lee R, Localio AR, Armstrong K, et al. A meta-analysis of the performance characteristics of the free prostate-specific antigen test. Urology. 2006;67(4):762-8. [Abstract]
- Carter HB, Partin AW, Luderer AA, et al. Percentage of free prostate-specific antigen in sera predicts aggressiveness of prostate cancer a decade before diagnosis. Urology. 1997;49(3):379-84. [Abstract]
- NCCN Clinical Practice Guidelines in Oncology: Prostate cancer early detection [online]. National Comprehensive Cancer Network; 1 June 2011 [cited 21 June 2011]. Available from: URL link
Dates
Tags
Created by:

 Login
Login















