- Introduction to the acquired immune system
- Activation of the acquired immune system
- Lymphocytes
Antibodies (immunoglobulins)
- The complement cascade
- Transplants, skin grafts and rejection
- Types of rejection
- Graft versus host reactions
- Immunity in babies
- Infections in the elderly
- Immunisation
Introduction to the acquired immune system
The human acquired immune system is responsible for the destruction of foreign particles once they have entered the body. Before it has seen a foreign particle, it is actually quite ignorant about how to destroy it. During the first exposure to an invader (which could be a virus, a bacteria or any unwanted particle), the acquired immune system must ‘learn’ how to attack and destroy the foreign particle. This means that it is not as good as the innate immune system for keeping out things that it has never encountered before. Once the acquired immune system has created a response, however, a protective response can be made more quickly and with greater force, allowing it to protect the body from harm.
The cells of the acquired immune system are mainly the B cells and the T cells, but there are also other important parts of the acquired immune system, such as the ‘complement cascade‘ and the production of antibodies. The acquired immune system also plays the key role in the rejection of implanted tissue.
Activation of the acquired immune system
Unlike the innate immune system, the acquired immune system needs to have seen a substance before in order to attack it effectively. This is because the way that the acquired immune system attacks a target is very specific and takes time to prepare.
All agents foreign to your body have unique patterns on their surfaces that allow the cells of the acquired immune system to detect them. When the cells of the aquired immune system detect these patterns, the agents are recognised as foreign, and the immune system can therefore mount an attack. Anything that the immune system can detect and attack is called an antigen.
The activation of the acquired immune system initially requires the help of other cells. The cells of the acquired immune system are coated in receptors. These are highly specific molecules designed to recognise certain substances. The receptors are so specific that each receptor can only recognise one substance and nothing else. There are many immune cells in the blood, each with its own different receptor. This means that the body can be protected against many different things.
Cells called macrophages (which means ‘big eater’) can speed up the process of activation. Macrophages are found in lots of places throughout the body, and eat anything that they do not recognise. After they have eaten something, the macrophages break it down into its basic proteins and present these to the immune cells. This causes a better, more accurate and more damaging response than the macrophages alone are capable of producing.
Lymphocytes
T cells: Mediated immunity
T cells account for about 80% of all lymphocytes. They are named T cells because they mature in the thymus, a gland found in the chest. There are three types of T cell lymphocytes:
- Cytotoxic T cells
- Suppressor T cells
- Helper T cells
These cells all play a role in the direct destruction of problem cells in the body, such as cells infected with a virus, or cells with DNA damage (e.g. some cancer cells).
T cell production
T cells start out as stem cells (early types of cells that have not yet fully grown) and are produced by bone marrow. To mature, these stem cells move to the thymus, where they can stay for up to three weeks. About 99% of T cells do not make it to maturity. This is because the body is very selective about what T cells are produced so that they do not cause damage to the body’s own cells. In the thymus, the T cells are given T cell receptors, of which there are several types. The type of receptor received determines what type of T cell it will be, what its role is, and which cells it can interact with.
T cell function
T cells function both through the release of substances into the blood, and by signalling B cells through contact. They have several different roles:
- Signalling for growth and activation of B cells
- Activation of cells that can ‘eat’ foreign substances
- Stimulation of cytotoxic T cells during a viral infection
- Signalling growth in cells, including other T cells, macrophages and eosinophils
T cell activation
T cells cannot detect foreign substances without assistance, and require a complex system to help them work. They need the help of cells called antigen presenting cells (APCs). These cells will eat the foreign substance, be it a bacteria, virus-infected cell or toxin, break it down, and present part of it to the T cell so that it can mount a response. APCs have a special type of molecule on their surface that allows them to communicate with helper T cells. Once a response is activated, lots and lots of T cells of different types are released into the blood stream. The released cells are responsible for the destruction of the foreign substance.
Helper T cells
Helper T cells are by far the most common T cell. They make up more than three quarters of the T cell population.Helper T cells help the immune system in many different ways, and serve as a major regulator of virtually all immune functions in the body. They mainly act through the release of substances that help control the other parts of the immune system. These substances (called lymphokines) stimulate the other types of T cells to grow and attack. They also help B cells grow and mature into their active form.In acquired immunodeficiency syndrome (AIDS), there is a loss of helper T cells, leaving the body open to infection. Also, due to the influence of helper T cells on B cells, B cells may be inactive in cases where the T cells are damaged.
Cytotoxic T cells
Cytotoxic T cells are important in defending against virally infected cells, in the rejection of tissue grafts, and in the immune response to certain tumour types. Cytotoxic T cells require activated APCs, and rely on the presence of helper T cells.
Following activation by helper T cells, cytotoxic T cells prepare for the destruction of their target. Inside the cells, substances are formed which are incredibly dangerous to cells. They create a protein called perforin, named because it has the ability to ‘perforate’ infected cells by punching holes in them. Cytotoxic T cells can also release enzymes that destroy the cell structure.
To destroy a cell, a cytotoxic T cell first latches on to it, then releases the aforementioned substances directly onto the cell. Consequently, there is no damage to any other cell that happens to be nearby. The released substances cause the cell to self-destruct rather than explode. Thus, if there are any viral particles in the cell, they will be destroyed with the cell rather than be released to spread. After the cell is destroyed, the cytotoxic T cell can detach itself and leave to destroy other infected or otherwise damaged cells.
Suppressor T cells
Suppressor T cells are, as the name suggests, capable of suppressing the functions of both helper and cytotoxic T cells. It is believed that suppressor cells function as regulators of the other cells of the immune system, stopping them from causing excessive damage to the body’s own tissues. It is probable that suppressor T cells play an important role in protecting against autoimmune attack.
B cells: Humoral immunity
B cell production
B cells start out as the same type of stem cell as the T cells. Instead of moving to the thymus, however, B cells move to bone marrow to mature. There they are given cell receptors, and are then released into the blood. Once released, they move to the lymphoid tissue of the body, where they are located nearby, but distinctly separate from, the T cells.
The production of B cells involves some incredibly complex genetics that is not worth speaking about here. Generally, imagine that the body has an enormous pile of building blocks with which to make something that will recognise a foreign substance. These building blocks can be placed together in millions of different combinations, and each will be able to recognise a certain substance. The body makes almost all possible combinations in the form of immunoglobulins and places them onto the surface of B cells, allowing them to recognise foreign substances.
B cell function
B cells play two major roles in the protection of the body:
- Ensuring antibody production against the appropriate target antigen; and
- Presenting antigens to T cells and providing signals for T cell activation.
B cell activation
The majority of B cell activation takes place in the lymph nodes. Certain types of cells in the lymph nodes eat anything foreign and present them to B and T cells. Any B cell that shares a receptor for this substance will be activated and start to multiply. B cells can also be activated by helper T cells. After activation, active B cells migrate around the body and change into plasma cells.
Plasma cells
Plasma cells are B cells that remain committed to the production and secretion of a single antibody type. This secretion gives rise to the antibodies found in the circulation. Immunity is kept for as long as the plasma cell continues to secrete antibodies.
Memory B cells
Memory B cells can also be formed after stimulation. These cells migrate to the lymph nodes, where they remain ready for further rounds of activation should the specific antigen ever be encountered again. If it is, then a very quick response can be made because memory B cells are ready and waiting to multiply.
Antibodies (immunoglobulins)
Antibodies are proteins that have the ability to bind antigens. While the terms ‘antibodies’ and ‘immunoglobulins’ mean slightly different things, they are often referred to interchangeably. Antibodies have many uses within the body, including:
- Targeting an infective organism
- Recruitment of immune cells
- Neutralisation of toxins
- Removal of foreign antigens from circulation
- Complement activation
- Stimulation of cells that can ‘eat’ foreign substances
- Activation of mast cells
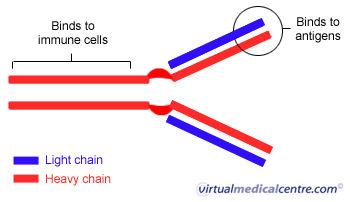
Structure
The typical immunoglobulin (Ig) is made up for four parts called ‘chains’. Two of the parts are slightly heavier, and so the Ig molecule is said to be made of two heavy chains and two light chains. These are arranged in a Y-shaped configuration. At the end of the two ‘arms’ is an area that has the ability to recognise foreign substances.
There are several different types of immunoglobulin.
Immunoglobulin G (IgG)
IgG is the most abundant immunoglobulin, and is one of the major activators of the complement pathway. Part of the IgG molecule is able to interact with lots of cells of the immune system, and so it has the ability to stimulate a very direct attack on anything that it recognises. IgG is also the only type of immunoglobulin that is transferred across the placenta from mother to foetus. This gives unborn children some protection from diseases to which they have not yet been exposed.
Immunoglobulin A (IgA)
IgA is the second most abundant immunoglobulin. An immunoglobulin (or antibody) is a protein which is involved in immune responses. IgA plays an important role in the cellular defence of mucosal surfaces. Mucosal surfaces are the moist, soft surfaces of the body (e.g. the inside of the mouth, all along the digestive system). IgA is excreted into bodily secretions such as tears, saliva and breast milk. A lack of IgA often causes major infections of mucosal surfaces, such as the mouth, throat and lungs.
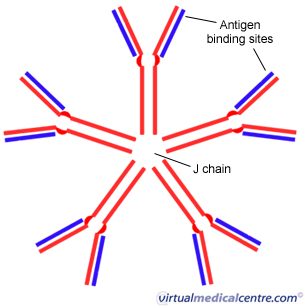
Immunoglobulin M (IgM)
IgM looks different than the other immunoglobulins, but is in fact just five normal immunoglobulins joined at their bases. IgM is the first immunoglobulin synthesised in an antibody response. It is a strong activator of the complement cascade.
Immunoglobulin E (IgE)
IgE is the largest immunoglobulin, but is present in extremely low levels in a healthy individual. IgE levels rise in response to parasitic infections and in individuals who are strongly allergic to something. The main action of IgE is binding to and activating mast cells. Mast cells cause local and occasionally generalised effects (e.g. swelling, redness, pain and itchiness). Hayfever is a condition caused by too much IgE activity.
The complement cascade
The complement system or cascade is a combination of 20 proteins that have the ability to directly destroy foreign cells. The complement pathway can be activated either as part of the innate immune system, or via the ‘classical pathway’ discussed here.
The classic pathway for complement activation involves the binding of an antibody to an antigen, whereby a foreign substance is coated with antibodies secreted from B cells. These antibodies bind directly to the ‘first’ complement enzyme. Multiple different substances are formed by this process. Each is designed to prevent damage to the body’s tissues while causing maximum damage to the invader.
Complement activation has the following effects on foreign substances (e.g. bacteria):
- Signalling and eating: Complement coats bacteria with signals that highlight it for the body’s ‘eaters’, leading to its destruction.
- Direct destruction: One substance formed can punch holes in bacteria walls, causing it to burst.
- Agglutination: Alteration of the bacterial surface by complement proteins cause them to stick to each other, making their destruction easier.
- Neutralisation of viruses: Complement proteins can directly attack viruses, rendering them harmless.
- Cell signalling: Complement causes neutrophils and macrophages to move next to antigens.
- Activation of mast cells and basophils: Activating mast cells and basophils causes the release of substances that lead to increased local blood flow, increased leakage of damaging proteins, and other factors that help inactivate or immobilise bacteria.
- Inflammation: Several complement proteins act to increase inflammation.
Transplants, skin grafts and rejection
Types of rejection
Hyperacute rejection
Hyperacute rejection is caused by preformed antibodies in the recipient that are directed against the cells from the donor. It occurs minutes to hours after transplantation. An example of this is when transplants are made across the ABO blood group barrier. If tissue from a group A donor is transplanted into an O patient, the O patient already has lots of antibodies against the A blood type within their circulation, and the foreign tissue is rejected. This type of rejection can also be due to the recipient having previously seen similar tissue before. An example of this is when white blood cells from a blood transfusion share some patterns with the recipient.
This form of rejection causes the formation of clots, and eventually the death of the implanted organ.
Acute rejection
Acute rejection is mediated by T cells and usually becomes apparent 7 days post-transplant. The recipient’s T cells recognise alloantigens and cause white blood cell infiltrations of the transplanted organ.
Chronic rejection
Chronic rejection appears months or years after successful transplantation, and is the major cause of long term graft loss. Blood vessel damage may predispose to chronic rejection, but the exact mechanism is poorly understood. It is mostly characterised by cell proliferation in the vascular walls and narrowing of the blood vessels, stopping adequate blood flow.
Graft versus host reactions
Graft versus host reactions arise when stem cells that can produce active immune cells are transplanted from a donor. It is most common following bone marrow transplantation, but has also been described following liver transplantation and blood transfusion. There are two forms of graft versus host reactions:
- Acute: Occurs within 1–2 months of a transplant
- Chronic: Develops 2–3 months post-transplant.
Graft versus host disease typically affects the liver, skin, intestinal tract and immune system within days or weeks of a bone marrow transplant. In its mild form, it can manifest as redness of the palms, soles and ears. There can also be signs of mild liver disease, and gastrointestinal signs such as mild diarrhoea. In more severe disease, there can be death of the skin, severe liver abnormalities and copious diarrhoea that can cause life-threatening dehydration.
Mild graft versus host disease may resolve spontaneously, or with immunosuppression. The severe form of the disease is usually resistant to treatment and has a fatal outcome.
Immunity in babies

After birth, when the mother is no longer passing antibodies to the child in the womb, the baby’s immunity can fall for a month or so. The antibodies from the mother can provide protection for about six months, and the baby will start creating antibodies well before then.
As has been mentioned before, the acquired immune system needs to encounter a foreign substance before it can mount an effective attack. This is why children seem to get more colds and other infections than adults. As children age and their immune systems encounter more and more infectious agents, they can fight infections more effectively.
Infections in the elderly
The elderly also have a decreased immune response, but while children get infections because their immune systems are young and hve not seen much, the reason is different for the elderly. As people get older, lots of their cells start to ‘slow down’ or become less effective. This includes the immune cells, and they are unable to mount as quick or as strong a response as a younger person.
The immune system can be affected by lots of other things besides age. Smoking makes people far more likely to get infections because it ‘slows’ the immune system. Some illnesses and medications can also cause the immune system to be less effective.
Immunisation
Immunisations use the acquired immune system to protect children (and adults) from many dangerous diseases. These diseases can be very serious and can lead to major problems for the infected children.Vaccines usually involve the injection of the whole or segments of the infectious agent; however, the injected agent is inactive. This means that the acquired immune system can see the foreign particles, attack them, and build up a proper defence against them without being exposed to the full infection. If the infectious agent is encountered, rather than having to spend time acquiring immunity (during which time major damage can occur), the body can attack and destroy it straight away.
More information
 |
For an overview of the immune system, see Human Immune System. |
 |
For information on the innate immune system, see Innate Immune System. |
References
- Martini, FH. Human Anatomy (Third Edition). New Jersey: Prentice-Hall; 2000.
- Guyton AC, Hall JE. Textbook of Medical Physiology. Philadelphia: Harcourt Health Sciences; 2000.
- Peakman M, Vergani D. Basic and Clinical Immunology. London: Churchill Livingstone (Elsevier); 2003.
- Starr C. Biology: Concepts and Applications (Fourth Edition). Pacific Grove: Brooks/Cole; 2000.
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.