Blood facts
- Approximately 8% of an adult’s body weight is made up of blood.
- Females have around 4-5 litres, while males have around 5-6 litres. This difference is mainly due to the differences in body size between men and women.
- Its mean temperature is 38 degrees Celcius.
- It has a pH of 7.35-7.45, making it slightly basic (less than 7 is considered acidic).
- Whole blood is about 4.5-5.5 times as viscous as water, indicating that it is more resistant to flow than water. This viscosity is vital to the function of blood because if blood flows too easily or with too much resistance, it can strain the heart and lead to severe cardiovascular problems.
- Blood in the arteries is a brighter red than blood in the veins because of the higher levels of oxygen found in the arteries.
- An artificial substitute for human blood has not been found.
Functions of blood

Transport
Blood transports the following substances:
- Gases, namely oxygen (O2) and carbon dioxide (CO2), between the lungs and rest of the body
- Nutrients from the digestive tract and storage sites to the rest of the body
- Waste products to be detoxified or removed by the liver and kidneys
- Hormones from the glands in which they are produced to their target cells
- Heat to the skin so as to help regulate body temperature
Protection
Blood has several roles in inflammation:
- Leukocytes, or white blood cells, destroy invading microorganisms and cancer cells
- Antibodies and other proteins destroy pathogenic substances
- Platelet factors initiate blood clotting and help minimise blood loss
Regulation
Blood helps regulate:
- pH by interacting with acids and bases
- Water balance by transferring water to and from tissues
Composition of blood
Blood is classified as a connective tissue and consists of two main components:
- Plasma, which is a clear extracellular fluid
- Formed elements, which are made up of the blood cells and platelets
The formed elements are so named because they are enclosed in a plasma membrane and have a definite structure and shape. All formed elements are cells except for the platelets, which are tiny fragments of bone marrow cells.
Formed elements are:
- Erythrocytes, also known as red blood cells (RBCs)
- Leukocytes, also known as white blood cells (WBCs)
- Platelets

Leukocytes are further classified into two subcategories called granulocytes which consist of neutrophils, eosinophils and basophils; and agranulocytes which consist of lymphocytes and monocytes.
The formed elements can be separated from plasma by centrifuge, where a blood sample is spun for a few minutes in a tube to separate its components according to their densities. RBCs are denser than plasma, and so become packed into the bottom of the tube to make up 45% of total volume. This volume is known as the haematocrit. WBCs and platelets form a narrow cream-coloured coat known as the buffy coat immediately above the RBCs. Finally, the plasma makes up the top of the tube, which is a pale yellow colour and contains just under 55% of the total volume.
Blood plasma
Blood plasma is a mixture of proteins, enzymes, nutrients, wastes, hormones and gases. The specific composition and function of its components are as follows:
These are the most abundant substance in plasma by weight and play a part in a variety of roles including clotting, defence and transport. Collectively, they serve several functions:
- They are an important reserve supply of amino acids for cell nutrition. Cells called macrophages in the liver, gut, spleen, lungs and lymphatic tissue can break down plasma proteins so as to release their amino acids. These amino acids are used by other cells to synthesise new products.
- Plasma proteins also serve as carriers for other molecules. Many types of small molecules bind to specific plasma proteins and are transported from the organs that absorb these proteins to other tissues for utilisation. The proteins also help to keep the blood slightly basic at a stable pH. They do this by functioning as weak bases themselves to bind excess H+ ions. By doing so, they remove excess H+ from the blood which keeps it slightly basic.
- The plasma proteins interact in specific ways to cause the blood to coagulate, which is part of the body’s response to injury to the blood vessels (also known as vascular injury), and helps protect against the loss of blood and invasion by foreign microorganisms and viruses.
- Plasma proteins govern the distribution of water between the blood and tissue fluid by producing what is known as a colloid osmotic pressure.
There are three major categories of plasma proteins, and each individual type of proteins has its own specific properties and functions in addition to their overall collective role:
- Albumins, which are the smallest and most abundant plasma proteins. Reductions in plasma albumin content can result in a loss of fluid from the blood and a gain of fluid in the interstitial space (space within the tissue), which may occur in nutritional, liver and kidney disease. Albumin also helps many substances dissolve in the plasma by binding to them, hence playing an important role in plasma transport of substances such as drugs, hormones and fatty acids.
- Globulins, which can be subdivided into three classes from smallest to largest in molecular weight into alpha, beta and gamma globulins. The globulins include high density lipoproteins (HDL), an alpha-1 globulin, and low density lipoproteins (LDL), a beta-1 globulin. HDL functions in lipid transport carrying fats to cells for use in energy metabolism, membrane reconstruction and hormone function. HDLs also appear to prevent cholesterol from invading and settling in the walls of arteries. LDL carries cholesterol and fats to tissues for use in manufacturing steroid hormones and building cell membranes, but it also favours the deposition of cholesterol in arterial walls and thus appears to play a role in disease of the blood vessels and heart. HDL and LDL therefore play important parts in the regulation of cholesterol and hence have a large impact on cardiovascular disease.
- Fibrinogen, which is a soluble precursor of a sticky protein called fibrin, which forms the framework of blood clot. Fibrin plays a key role in coagulation of blood, which is discussed later in this article under Platelets.
These are formed from the break down of tissue proteins or from the digestion of digested proteins.
Being toxic end products of the break down of substances in the body, these are usually cleared from the bloodstream and are excreted by the kidneys at a rate that balances their production.
Those absorbed by the digestive tract are transported in the blood plasma. These include glucose, amino acids, fats, cholesterol, phospholipids, vitamins and minerals.
Some oxygen and carbon dioxide are transported by plasma. Plasma also contains a substantial amount of dissolved nitrogen.
The most abundant of these are sodium ions, which account for more of the blood’s osmolarity than any other solute.
Red blood cells
- To pick up oxygen from the lungs and deliver it to tissues elsewhere
- To pick up carbon dioxide from other tissues and unload it in the lungs
An erythrocyte is a disc-shaped cell with a thick rim and a thin sunken centre. The plasma membrane of a mature RBC has glycoproteins and glycolipids that determine a person’s blood type. On its inner surface are two proteins called spectrin and actin that give the membrane resilience and durability. This allows the RBCs to stretch, bend and fold as they squeeze through small blood vessels, and to spring back to their original shape as they pass through larger vessels.
RBCs are incapable of aerobic respiration, preventing them from consuming the oxygen they transport because they lose nearly all their inner cellular components during maturation. The inner cellular components lost include their mitochondria, which normally provide energy to a cell, and their nucleus, which contains the genetic material of the cell and enable it to repair itself. The lack of a nucleus means that RBCs are unable to repair themselves. However, the resulting biconcave shape is that the cell has a greater ratio of surface area to volume, enabling O2 and CO2 to diffuse quickly to and from Hb.
The cytoplasm of a RBC consists mainly of a 33% solution of haemoglobin (Hb), which gives RBCs their red colour. Haemoglobin carries most of the oxygen and some of the carbon dioxide transported by the blood.
Circulating erythrocytes live for about 120 days. As a RBC ages, its membrane grows increasingly fragile. Without key organelles such as a nucleus or ribosomes, RBCs cannot repair themselves. Many RBCs die in the spleen, where they become trapped in narrow channels, broken up and destroyed. Haemolysis refers to the rupture of RBCs, where haemoglobin is released leaving empty plasma membranes which are easily digested by cells known as macrophages in the liver and spleen. The Hb is then further broken down into its different components and either recycled in the body for further use or disposed of.
White blood cells
White blood cells (WBCs) are also known as leukocytes. They can be divided into granulocytes and agranulocytes. The former have cytoplasms that contain organelles that appear as coloured granules through light microscopy, hence their name. Granulocytes consist of neutrophils, eosinophils and basophils. In contrast, agranulocytes do not contain granules. They consist of lymphocytes and monocytes.
- Neutrophils: These contain very fine cytoplasmic granules that can be seen under a light microscope. Neutrophils are also called polymorphonuclear (PMN) because they have a variety of nuclear shapes. They play roles in the destruction of bacteria and the release of chemicals that kill or inhibit the growth of bacteria.
- Eosinophils: These have large granules and a prominent nucleus that is divided into two lobes. They function in the destruction of allergens and inflammatory chemicals, and release enzymes that disable parasites.
- Basophils: They have a pale nucleus that is usually hidden by granules. They secrete histamine which increases tissue blood flow via dilating the blood vessels, and also secrete heparin which is an anticoagulant that promotes mobility of other WBCs by preventing clotting.
- Lymphocytes: These are usually classified as small, medium or large. Medium and large lymphocytes are generally seen mainly in fibrous connective tissue and only occasionally in the circulation bloodstream. Lymphocytes function in destroying cancer cells, cells infected by viruses, and foreign invading cells. In addition, they present antigens to activate other cells of the immune system. They also coordinate the actions of other immune cells, secrete antibodies and serve in immune memory.
- Monocytes: They are the largest of the formed elements. Their cytoplasm tends to be abundant and relatively clear. They function in differentiating into macrophages, which are large phagocytic cells, and digest pathogens, dead neutrophils, and the debris of dead cells. Like lymphocytes, they also present antigens to activate other immune cells.
Platelets
Platelets are small fragments of bone marrow cells and are therefore not really classified as cells themselves.
Platelets have the following functions:
- Secrete vasoconstrictors which constrict blood vessels, causing vascular spasms in broken blood vessels
- Form temporary platelet plugs to stop bleeding
- Secrete procoagulants (clotting factors) to promote blood clotting
- Dissolve blood clots when they are no longer needed
- Digest and destroy bacteria
- Secrete chemicals that attract neutrophils and monocytes to sites of inflammation
- Secrete growth factors to maintain the linings of blood vessels
The first three functions listed above refer to important haemostatic mechanisms in which platelets play a role in during bleeding: vascular spasms, platelet plug formation and blood clotting (coagulation).
This is a prompt constriction of the broken blood vessel and is the most immediate protection against blood loss. Injury stimulates pain receptors. Some of these receptors directly innervate nearby blood vessels and cause them to constrict. After a few minutes, other mechanisms take over. Injury to the smooth muscle of the blood vessel itself causes a longer-lasting vasoconstriction where platelets release a chemical vasoconstrictor called serotonin. This maintains vascular spasm long enough for the other haemostatic mechanisms to come into play.
Under normal conditions, platelets do not usually adhere to the wall of undamaged blood vessels, since the vessel lining tends to be smooth and coated with a platelet repellent. When a vessel is broken, platelets put out long spiny extensions to adhere to the vessel wall as well as to other platelets. These extensions then contract and draw the walls of the vessel together. The mass of platelets formed is known as a platelet plug, and can reduce or stop minor bleeding.
This is the last and most effective defence against bleeding. During bleeding, it is important for the blood to clot quickly to minimise blood loss, but it is equally important for blood not to clot in undamaged vessels. Coagulation is a very complex process aimed at clotting the blood at appropriate amounts. The objective of coagulation is to convert plasma protein fibrinogen into fibrin, which is a sticky protein that adheres to the walls of a vessel. Blood cells and platelets become stuck to fibrin, and the resulting mass helps to seal the break in the blood vessel. The forming of fibrin is what makes coagulation so complicated, as it involved numerous chemicals reactions and many coagulation factors.
Production of blood
Haemopoiesis
Haemopoiesis is the production of the formed elements of blood. Haemopoietic tissues refer to the tissues that produce blood. The earliest haemopoietic tissue to develop is the yolk sac, which also functions in the transfer of yolk nutrients of the embryo. In the foetus, blood cells are produced by the bone marrow, liver, spleen and thymus. This changes during and after birth. The liver stops producing blood cells around the time of birth, while the spleen stops producing them soon after birth but continues to produce lymphocytes for life. From infancy onwards, all formed elements are produced in the red bone marrow. Lymphocytes are additionally produced in lymphoid tissues and organs widely distributed in the body, including the thymus, tonsils, lymph nodes, spleen and patches of lymphoid tissues in the intestine.
Erythropoiesis refers specifically to the production of erythrocytes or red blood cells (RBCs). These are formed through the following sequence of cell transformations:
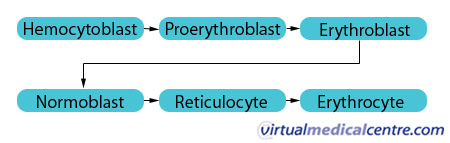
The proerythroblast has receptors for the hormone erythropoietin (EPO). Once EPO receptors are in place, the cell is committed to exclusively producing RBCs. The erythroblasts then multiply and synthesise haemoglobin (Hb), which is a red oxygen transport protein. The nucleus from the erythroblasts is then discarded, giving rise to cells named reticulocytes. The overall transformation from haemocytoblast to reticulocytes involves a reduction in cell size, an increase in cell number, the synthesis of haemoglobin, and the loss of the cell nucleus. These reticulocytes leave the bone marrow and enter the bloodstream where they mature into erythrocytes when their endoplasmic reticulum disappears.
Leukopoiesis refers to the production of leukocytes (WBCs). It begins when some types of haemocytoblasts differentiate into three types of committed cells:
- B progenitors, which are destined to become B lymphocytes
- T progenitors, which become T lymphocytes
- Granulocyte-macrophage colony-forming units, which become granulocytes and monocytes
These cells have receptors for colony-stimulating factors (CSFs). Each CSF stimulates a different WBC type to develop in response to specific needs. Mature lymphocytes and macrophages secrete several types of CSFs in response to infections and other immune challenges. The red bone marrow stores granulocytes and monocytes until they are needed in the bloodstream. However, circulating leukocytes do not stay in the blood for very long. Granulocytes circulate for 4-8 hours and then migrate into the tissues where they live for another 4-5 days. Monocytes travel in the blood for 10-20 hours, then migrate into the tissues and transform into a variety of macrophages which can live as long as a few years. Lymphocytes are responsible for long-tern immunity and can survive from a few weeks to decades. They are continually recycled from blood to tissue fluid to lymph and finally back to the blood.
Thrombopoiesis refers to the production of platelets in the blood, because platelets used to be called thrombocytes. This starts when a haemocytoblast develops receptors for the hormone thrombopoietin which is produced by the liver and kidneys. When these receptors are in place, the haemocytoblast becomes a committed cell called a megakaryoblast. This replicates its DNA, producing a large cell called a megakaryocyte, which breaks up into tiny fragments that enter the bloodstream. About 25-40% of the platelets are stored in the spleen and released as needed. The remainder circulate freely in the blood are live for about 10 days.
Ageing changes in the blood
The properties of blood change as we grow older. It is thought that these changes might contribute to the increased incident of clot formation and atherosclerosis in older people. Some of the most prominent findings on these changes include:
- Rise in fibrinogen
- Rise in blood viscosity
- Rise in plasma viscosity
- Increased red blood cell rigidity
- Increased formation of fibrin degradation products
- Earlier activation of the coagulation system
The increased level of plasma fibrinogen is thought to be due to either its rapid production or slower degradation. As age progresses, fibrinogen and plasma viscosity tend to be positively correlated, with the rise in plasma viscosity being largely attributed to the rise in fibrinogen.
The viscosity of blood depends on factors such as shear rate, haemocrit, red cell deformability, plasma viscosity and red cell aggregation. Although there are many factors involved, hyperviscosity syndrome can be generated by a rise in only one factor. A state of hyperviscosity causes sluggish blood flow and reduced oxygen supply to the tissue.
An age-dependent increase in various coagulation factors, a positive correlation with fibrinogen and a negative correlation with plasma albumin has also been found. Both platelet and red cell aggregation increase with age, with red cell aggregation appearing to be the primary factor responsible for a rise in blood viscosity at low shear rates.
The decrease in red cell deformability (increase in rigidity) refers to its ability to deform under flow forces. Less deformable cells offer more resistance to flow in the microcirculation, which influences the delivery of oxygen to the tissues. Studies have found that older people have less fluid membranes in their red cells.
Blood H+ has also been found to be positively correlated with age, making the blood slightly more acidic as we age. This results in a swelling of the cell, making the red cells less deformable. This sets up a cycle for further increase in blood viscosity and worsening of blood flow parameters.
Since ageing causes a reduction in total body water, blood volume decreases due to less fluid being present in the bloodstream. The number of red blood cells, and the corresponding haemoglobin and haemocrit levels, are reduced which contributes to fatigue in the individual. Most of the white blood cells stay at their original levels, although there is a decrease in lymphocyte number and ability to fight off bacteria, leading to a reduced ability to resist infection.
Overall, the rise in fibrinogen is the most common and significant change in blood during ageing because it contributes to a rise in plasma viscosity, red blood cell aggregation and a rise in blood viscosity at low shear rates. Increased age is associated with a state of hypercoagulation of blood, making older people more susceptible to clot formation and atherosclerosis.
More information
 |
For more information on blood, blood types, blood tests, and blood donation and transfusion, see Blood. |
References
- Ajmani RS, Rifkind JM. Hemorheological changes during human aging. Gerontology 1998; 44 (2): 111-120
- Coagulation cascade [online]. 2003 [cited 2007 Sep 9]. Available from: URL: http://labtestsonline.org/ understanding/ analytes/ coag_cascade/ coagulation_cascade.html
- Marieb EN. Human anatomy & physiology. 4th ed. Menlo Park, Calif.: Benjamin/Cummings; 1998.
- Saladin KS. Anatomy and physiology – the unity of form and function. 3rd ed. New York: McGraw-Hill; 2004.
- Sherwood L. Human physiology – from cells to systems. 5th ed. Belmont, Calif: Brooks/Cole; 2004.
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.